Cartilage, tendons, ligaments & skin
The tissues of cartilage, tendons and ligaments have strong common features. In scientific language they are part of the “extra cellular matrix” and in everyday life they are known as “connective tissue”. The building blocks of connective tissue are the same for cartilage, tendons, ligaments and skin (especially in the subcutaneous layers, the hypodermis): collagen fibres, elastin fibres and proteoglycans.
Structure proteins
Collagen is a protein only found in animals and humans. Plants do not hold any. Collagen is the most common protein in the human body and is constituted of amino acids assembled in a precise sequence. Collagen fibres are very resistant to pull and hardly stretch. They provide resistance and strength to tissues. For this reason, they are often termed as « structure proteins ». There are different sub-groups of collagens: types 1 and 3 are mainly found in skin, tendons, ligaments, bones and teeth, whereas collagen type 2 is mainly found in articular cartilage (hyaline and elastic cartilage).
Collagen is made up of three long chains of proteins which hold on to one another through disulfide and hydrogen bonds. Together these three long protein chains form a triple helix collagen, or procollagen (see image).
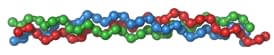
Illustration: Collagen triple-helix
Many collagen triple-helix form a fibril and many fibrils form a collagen fibre. We can picture a collagen fibre as a huge steel cable, like the extremely resistant cables used by cable cars.
Amino acids
The smallest building blocks of collagen and elastin fibres are amino acids. All in all, there are twenty different amino acids which are found in all proteins of the human body. Of these twenty amino acids, eight are essential, meaning that the human body does not create them (isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophane, Valine). When essential amino acids are not sufficiently available, the other amino acids cannot be used for the creation of new proteins and will be transformed into sugar and fat. This is why the human body needs a sufficient and regular intake of essential amino acids from food.
In collagen, the following amino acids are particularly present: glycine, proline, hydroxyproline and hydroxylysine. Threonine, an essential amino acid, is a forerunner for glycine synthesis.
Proteoglycans
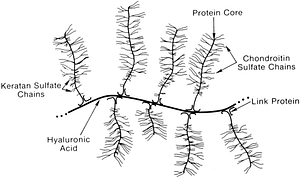 Proteoglycans are the third important element in connective tissues, particularly in cartilage. Proteoglycans are negatively charged and can bind water molecules. They confer the typical gelatinous consistency to connective tissue. Proteoglycans are situated between collagen and elastin fibres and enable the movement of molecules through the matrix, including feeding the tissues around them. We can picture the shape of proteoglycans like a fir tree: the trunk is made of hyaluronic acid, branches would be chondroitin, glucosamine, dermatan and keratane sulphates (see illustration).
Proteoglycans are the third important element in connective tissues, particularly in cartilage. Proteoglycans are negatively charged and can bind water molecules. They confer the typical gelatinous consistency to connective tissue. Proteoglycans are situated between collagen and elastin fibres and enable the movement of molecules through the matrix, including feeding the tissues around them. We can picture the shape of proteoglycans like a fir tree: the trunk is made of hyaluronic acid, branches would be chondroitin, glucosamine, dermatan and keratane sulphates (see illustration).
Illustration: Proteoglycan
With age, synthesis of collagen, elastin and proteoglycans (particularly chondroitine sulphates) decrease naturally. Productive cells (chondroblasts and fibroblast) become less productive with age.

